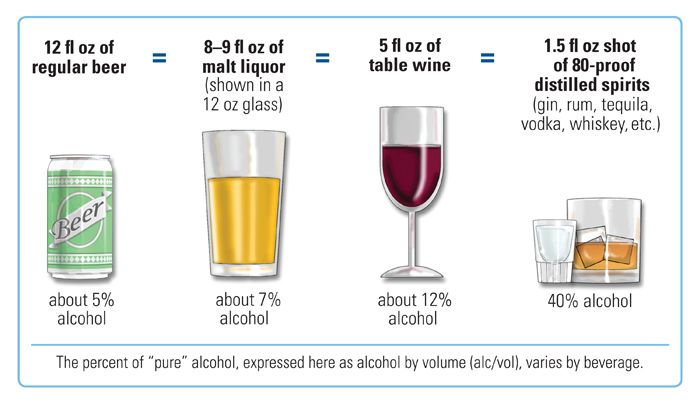
Standard Drink
In the United States, a standard drink is defined as 0.6 fluid ounce (18 mL or 14 g) of pure alcohol.[4] In the United States, therefore, one standard drink is contained in:
- a 12-ounce bottle or can of typical (5% ABV) beer
- a 5-ounce glass of typical (12% ABV) wine
- a 1.5-ounce shot of typical (80-proof, 40% ABV) liquor (either straight or in a mixed drink)
Alcohol equivalence is emphasized as being important for drinking in moderation by the National Institute on Alcohol Abuse and Alcoholism(NIAAA), Department of Transportation (DOT), Department of Health and Human Services (DHHS), American Dietetic Association, National Consumers League, National Council on Alcoholism and Drug Dependence (NCADD), and Mothers Against Drunk Driving (MADD).
http://www.niaaa.nih.gov/alcohol-health/overview-alcohol-consumption/what-standard-drink
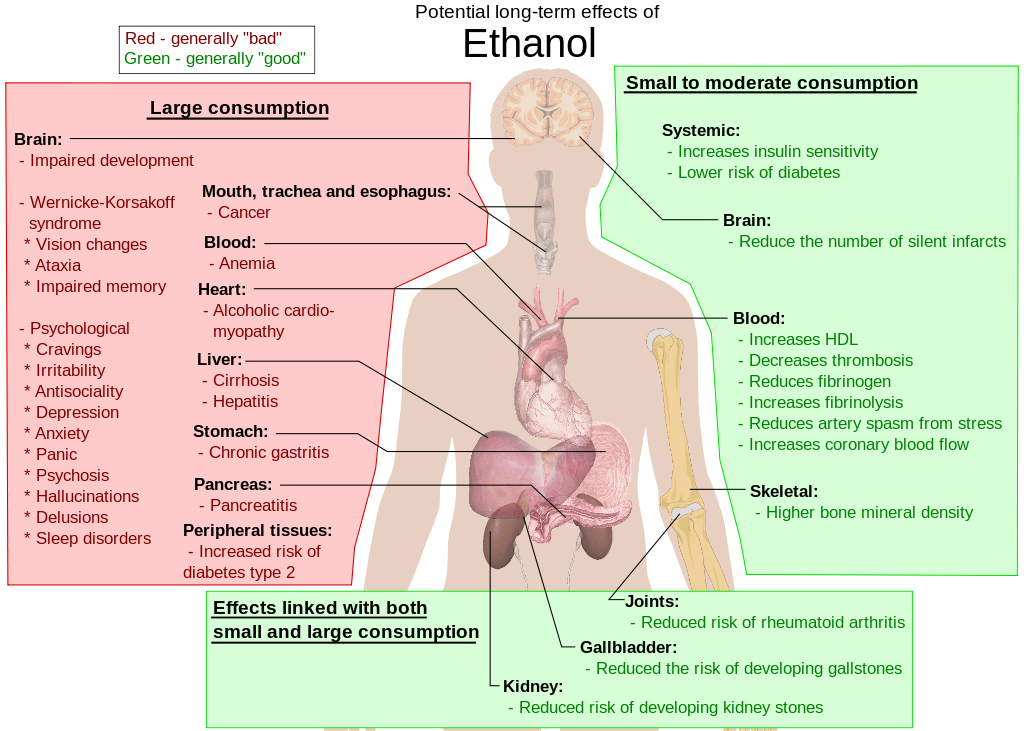
Long-term Effects of Alcohol
From: https://en.wikipedia.org/wiki/Long-term_effects_of_alcohol_consumption
The long-term effects of alcohol (ethanol) consumption range fromcardioprotective health benefits for low to moderate alcohol consumption in industrialized societies with higher rates of cardiovascular disease[1][2] to severe detrimental effects in cases of chronic alcohol abuse.[3] Health effects associated with alcohol intake in large amounts include an increased risk of alcoholism, malnutrition,chronic pancreatitis, alcoholic liver disease, and cancer. In addition, damage to the central nervous system and peripheral nervous systemcan occur from chronic alcohol abuse.[4][5] The long-term use of alcohol is capable of damaging nearly every organ and system in the body.[6]The developing adolescent brain is particularly vulnerable to the toxic effects of alcohol.[7] In addition, the developing fetal brain is also vulnerable, and fetal alcohol spectrum disorders (FASDs) may result if pregnant mothers consume alcohol.
The inverse relation in Western cultures between alcohol consumption and cardiovascular disease has been known for over 100 years.[8]Many physicians do not promote alcohol consumption, however, given the many health concerns associated with it, some suggest that alcohol should be regarded as a recreational drug, and promote exercise and good nutrition to combat cardiovascular disease.[9][10] Others have argued that the benefits of moderate alcohol consumption may be outweighed by other increased risks, including those of injuries,violence, fetal damage, liver disease, and certain forms of cancer.[11]
Withdrawal effects and dependence are also almost identical.[12]Alcohol at moderate levels has some positive and negative effects on health. The negative effects include increased risk of liver diseases,oropharyngeal cancer, esophageal cancer and pancreatitis. Conversely moderate intake of alcohol may have some beneficial effects ongastritis and cholelithiasis.[13] Of the total number of deaths and diseases caused by alcohol, most happen to the majority of the population who are moderate drinkers, rather than the heavy drinker minority.[14] Chronic alcohol misuse and abuse has serious effects on physical and mental health. Chronic excess alcohol intake, or alcohol dependence, can lead to a wide range of neuropsychiatric orneurological impairment, cardiovascular disease, liver disease, andmalignant neoplasms. The psychiatric disorders which are associated with alcoholism include major depression, dysthymia, mania,hypomania, panic disorder, phobias, generalized anxiety disorder,personality disorders, schizophrenia, suicide, neurologic deficits (e.g. impairments of working memory, emotions, executive functions,visuospatial abilities and gait and balance) and brain damage. Alcohol dependence is associated with hypertension, coronary heart disease, and ischemic stroke, cancer of the respiratory system, and also cancersof the digestive system, liver, breast and ovaries. Heavy drinking is associated with liver disease, such as cirrhosis.[15] Excessive alcohol consumption can have a negative impact on aging.[16]
Recent studies have focused on understanding the mechanisms by which moderate alcohol consumption confers cardiovascular benefit.[17]
Binge Drinking
From: https://en.wikipedia.org/wiki/Binge_drinking
Binge drinking or heavy episodic drinking is a modern epithet for drinking alcoholic beverages with an intention of becoming intoxicated by heavy consumption of alcohol over a short period of time.[1] It is a style of drinking that is popular in several countries worldwide, and overlaps somewhat with social drinking since it is often done in groups. The degree of intoxication, however, varies between and within various cultures that engage in this practice. A binge on alcohol can occur over hours, last up to several days, or in the event of extended abuse, even weeks. Due to the long-term effects of alcoholmisuse, binge drinking is considered to be a major public health issue.[2]
Binge drinking is associated with a profound social harm, economic costs as well as increased disease burden. Binge drinking is more common in males, during adolescence and young adulthood. Heavy regular binge drinking is associated with adverse effects on neurologic,cardiac, gastrointestinal, hematologic, immune, musculoskeletal organ systems as well as increasing the risk of alcohol induced psychiatric disorders.[3][4] A US-based review of the literature found that up to one-third of adolescents binge-drink, with six percent reaching the threshold of having an alcohol-related substance use disorder.[5] Approximately one in 25 women binge-drinks during pregnancy, which can lead to fetal alcohol syndrome and fetal alcohol spectrum disorders.[6] Binge drinking during adolescence is associated with traffic accidents and other types of accidents, violent behavior as well as suicide. The more often a child or adolescent binge drinks and the younger they are the more likely that they will develop an alcohol use disorder includingalcoholism. A large number of adolescents who binge-drink also consume other psychotropic substances.[7]
Binge drinking causes brain damage faster and more severely than chronic drinking (alcoholism). The neurotoxic insults are due to very large amounts of glutamate which are released and over-stimulate the brain as a binge finishes. This results in excitotoxicity, a process which damages or kills neurons (brain cells).[8] Each binge drinking episode immediately insults the brain; repeat episodes result in accumulating harm. The developing adolescent brain is thought to be particularly susceptible to the neurotoxic effects of binge drinking, with some evidence of brain damage occurring from drinking more than 10 or 11 drinks once or twice per month.[9]
Alcohol Metabolism
Ethanol Consumption
As a person starts drinking alcohol, up to 5% of the ingested ethanol is directly absorbed and metabolized by some of cells of the gastrointestinal tract (the mouth, tongue, esophagus and stomach). Up to 100% of the remaining ethanol travels in circulation. This is one reason why blood tests are more accurate in measuring alcohol levels.
The lungs and kidneys will excrete about 2% to 10% of this circulatory ethanol. The more you drink the more quick trips to the restroom. The human body dehydrates as a result of these frequent trips to the restroom. This dehydration affects every single cell in your body, including your brain cells. This is the cause of the so-called “morning hangover”. Do not take Tylenol (acetaminophen). Alcohol metabolism activates an enzyme that transforms acetaminophen into a toxic metabolite that causes liver inflammation and damage. Liver damage may not be irreversible. Instead, drink water with electrolytes or sport drinks to rehydrate the body’s cells.
Alcohol is a volatile (flammable) organic substance and can be converted to a gas. The lungs exhale alcohol as a gas. The more alcohol consumed, the stronger the smell of alcohol in a person’s breathe. Breathalyzer tests measure the exhaled alcohol levels in the lungs to determine the state of inebriation.
The liver metabolizes up to 85% – 98% of the circulatory ethanol. The liver uses two metabolic processes to get rid of this circulatory ethanol as quickly and safely as possible.
- Alcohol dehydrogenase system
- Microsomal ethanol oxidizing system (MEOS)
Alcohol Dehydrogenase System
About 80 to 90% of the total hepatic ethanol uptake is processed via the alcohol dehydrogenase system. The reminder 10 to 20% of the total hepatic ethanol uptake is processed via the microsomal ethanol oxidizing system (MEOS).
The degradation of ethanol begins in the cytosol of the hepatic cell. The main cytosolic enzyme that catalyzes this reaction is called alcohol dehydrogenase. The products from this reaction are acetaldehyde, NADH and H+ ion. Acetaldehyde is very toxic to the liver and the body’s cells. The moment acetaldehyde is produced; it must be degraded to protect the liver cells. The enzyme that will carry this type of degradation reaction is acetaldehyde dehydrogenase (ALDH). Acetaldehyde dehydrogenase is found in both the mitochondria (ALDH2) and the cytosol (ALDH1) of the hepatic cells. Most of the hepatic acetaldehyde is then transported to the mitochondria for further degradation. A small portion is degraded in the cytosol of the hepatic cells. Acetaldehyde dehydrogenase converts acetaldehyde into acetate, a non-toxic molecule. Other products from this reaction are NADH and H+ ion.
Microsomal Ethanol Oxidizing System (MEOS)
In a moderate drinker, about 10 to 20% of the total hepatic ethanol uptake is processed via the microsomal ethanol oxidizing system (MEOS). During periods of heavy drinking, the MEOS system will metabolize most of the excess ethanol ingested. Heavy drinking stimulates the synthesis of all cytochrome P450 enzymes in the human body to include the MEOS system enzymes. This will help clear ethanol faster from the body.
The MEOS system is located on the membrane of the endoplasmic reticulum (inside the cytosol of the hepatic cell) and contains a cytochrome P450 enzyme complex. The enzyme complex is made up of two subunits: a cytochrome P450 reductase and a cytochrome P450 (also known as CYP2E1). Oxygen gas (O2 ) binds the iron-heme of the cytochrome P450 (CYP2E1). This activates the cytochrome P450 (CYP2E1) to accept electrons from NADPH. The cytochrome P450 reductase first transfers these two electrons from NADPH to the iron-heme of the cytochrome P450 (CYP2E1). Ethanol binds the other binding site of cytochrome P450 (CYP2E1) and is converted to acetaldehyde. This acetaldehyde is then further degraded by cytosolic (ALDH1) and mitochondrial (ALDH2) acetaldehyde dehydrogenase. Acetaldehyde dehydrogenase immediately converts acetaldehyde into acetate, a non-toxic molecule. Other products from this reaction are NADH and H+ ion.
Fate of Acetate
The acetate produced (from the alcohol dehydrogenase system and microsomal ethanol oxidizing system) is either released into circulation (major) or retained inside the liver cells (minor).
In the cytosol of the hepatic cells, acetate is converted to acetyl CoA. The enzyme responsible for this reaction is acetyl CoA synthetase. The acetyl CoA can:
- Enter the TCA cycle to produce CO2, FADH2, three molecules of NADH and three H+ ions.
- Be used in the synthesis of fatty acids and cholesterol.
In circulation, acetate is taken up by skeletal muscle, heart muscle and other tissue cells. Acetate is converted to acetyl CoA. The enzyme responsible for this reaction is acetyl CoA synthetase. Acetyl CoA enters the TCA cycle to produce CO2, FADH2, three molecules of NADH and three H+ ions. As the levels of CO2 begin to increase, the blood pH begins to decrease.
Excess Acetaldehyde and Generation of Radicals
A small amount (up to 10%) of the hepatic acetaldehyde may accumulate inside the liver cells. As more alcohol is ingested, this stimulates both the synthesis of the cytochrome P450 enzymes in the MEOS system and the production of acetaldehyde by both the alcohol dehydrogenase and microsomal ethanol oxidizing systems. The mitochondrial enzyme acetaldehyde dehydrogenase cannot convert this excess acetaldehyde into acetate as fast as it is produced.
Excess cytochrome P450 enzyme complexes can also generate radicals in the cytosol of the hepatic cells with increasing alcohol consumption. Ethanol induces the generation of free radicals from both the cytochrome P450 reductase and cytochrome P450 (CYP2E1) enzymes. The free radicals are called hydroxyethyl (CH3 CH2 O∙) radicals.
As the levels of acetaldehyde and radicals (CH3 CH2 O∙) increase inside the liver cells with heavy consumption of alcohol, some of the acetaldehyde and free radicals diffuse into circulation. In circulation, high levels of acetaldehyde cause nausea and vomiting. Vomiting causes more body dehydration and loss of electrolytes. If the dehydration becomes severe enough, this can impair brain function and a person may lose consciousness. As the body continues to lose water volume, the blood becomes more concentrated with alcohol and acetaldehyde resulting in alcohol intoxication, a lethal condition. A person should seek medical attention immediately to prevent death.
Accumulation of both acetaldehyde and free radicals (CH3 CH2 O∙) causes damage to the body’s cells. Acetaldehyde binds amino acid side groups, sulfhydryl groups, nucleotides (RNA & DNA bases) and phospholipids (cell membrane components) in the human body. It affects all the proteins in the human body to include proteins in the liver and the heart. In the liver, it affects the release of plasma proteins (albumin, blood coagulation factors, and transport proteins) and VLDL particles into circulation. Accumulation of both plasma proteins and lipid draws water inside the hepatic cell. Swelling of the hepatic cells impairs cell function and blood flow via the hepatic portal vein (portal hypertension).
Acetaldehyde also binds antioxidant systems in the body to include glutathione and free-radical defense enzymes. This enhances the formation of more radicals in the human body and the activation of potential toxins and carcinogens. Excess radicals generated from both acetaldehyde binding to antioxidant systems and the induction of the MEOS system cytochrome P450 enzymes will attack phospholipids in any cell membrane to include mitochondrial membranes. In the liver, peroxidation of the phospholipids in the inner mitochondrial membrane causes:
- Inhibition of the electron transport chain and uncoupling of oxidative phosphorylation. This causes inflammation and cell necrosis as a result of no ATP synthesis.
- Inhibition of β-oxidation of fatty acids. Increase synthesis and storage of lipids in the cytosol of the hepatic cells
- Damage to the alcohol dehydrogenase system as the mitochondria degrades. This leads to increase build up of both acetaldehyde and radicals by the MEOS system.
- The urea cycle is affected as the mitochondrion degrades. This releases toxic levels of ammonia into circulation (hyperammonemia).
- As the cells begin to rupture or die, bilirubin cannot be conjugated inside the liver cells. Bilirubin is released into circulation and accumulates inside the lipid membranes of the cells. This condition is known as jaundice.
Any of these effects may result in cirrhosis of the liver, hepatocarcinomas or alcohol-induced hepatitis. The transport of both acetaldehyde and free radicals (CH3 CH2 O∙) in circulation causes damage to other cells in the peripheral tissues. This may cause lung or breast cancer.